|
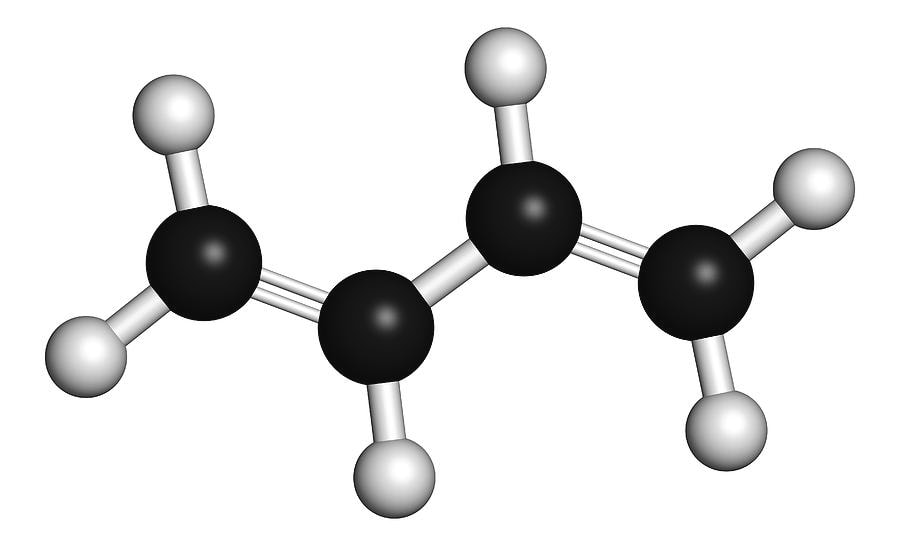
Butadiene is a colorless, odorless gas, which is used in the production of synthetic rubber. Its structure can be viewed as a union of two vinyl groups. When vaporized, it is easily condensed into liquid form. This compound is important in the industrial sector as a precursor to synthetic rubber.
Styrene-Butadiene Rubber (SBR) is a family of synthetic rubbers. These compounds have good abrasion and aging resistance and are produced through the process of copolymerizing styrene with butadiene. Butadiene is a very important building block in synthetic rubber. Its use is particularly prevalent in the tire industry. There are two major end uses of butadiene: replacement and original equipment (OE). The RE segment is largely driven by automotive production. Butadiene is used in the manufacturing of a number of products, including tires and polystyrene. It is often used in blends to reduce rolling resistance and improve resilience. However, the demand for butadiene is weak. Demand for Butadiene has decreased over the past few months. While global demand for butadiene has rebounded, Chinese butadiene has been weak since February. Despite this, China's butadiene market is expected to recover during the next few years. Due to a lack of profitability, on-purpose butadiene producers are unable to maintain their operating rates. They are also struggling to find raw materials. In some countries, the price of butene-1 and butene-2 feedstock is so high that new on-purpose butadiene capacities have been delayed. Butadiene polymerization is a reaction of Butadiene with a catalyst system in a hydrocarbon solvent. It is known to produce a polymer with a high Mooney viscosity. This type of polymerization can be achieved in the presence of a cobalt compound or a Mooney viscosity jump agent. The aging period and the type of Mooney viscosity jump agent used will have an effect on the rise in the polymer's viscosity. In butadiene polymerization, a secondary alkyl chloride may be used as the initiator. A high molecular weight polybutadiene is usually obtained by polymerizing Butadiene in the presence of a metallocene catalyst. These are effective catalysts for high cis polymers with broad molecular weights. They are also efficient catalysts for the synthesis of stereospecific high-cis polymers. As the polymerization reaction proceeds, the concentration of the polymer increases. This is because a new monomer is added to the previous butadiene unit. During this process, a new double bond is formed between the two carbons of the new butadiene unit. Depending on the structure of the newly formed butadiene polymer, it may be cis or trans. However, a common polar additive, a superbase, can affect the microstructure. Polybutadiene polymerization is carried out at temperatures between -30 C and +150 C. After a certain period of time, the catalyst system is removed from the solution. Typically, a concentration of 15 percent of charged monomer is maintained. Butadiene is a ubiquitous environmental pollutant. It is found in the smoke from tobacco as well as from automobile exhaust. And it is a key component in the production of synthetic rubber products. Depending on the amount of exposure, butadiene can cause different health effects. For example, it can irritate the eyes, throat, and nasal passages. The best way to protect from the dangers of butadiene is to store it in a cool, well-ventilated area. 1,3-butadiene is a colorless, gaseous chemical. It is also the most carcinogenic compound in cigarette smoke. This is because it is formed in the incomplete combustion of fossil fuels. Despite its carcinogenic nature, it is still a common petrochemical.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|
 RSS Feed
RSS Feed
