|
Formic Acid is also known as methanoic acid, is the simplest carboxylic acid. It is a common industrial reagent and occurs naturally in stinging insects, plants, unripe fruit and muscle tissue.
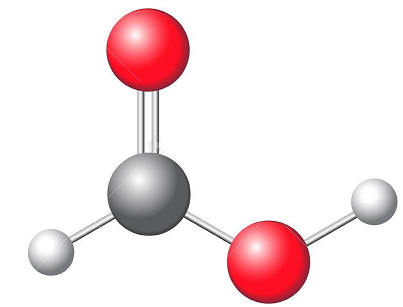
Methanoic acid is produced as a byproduct of acetic acid manufacture. It is also prepared on a large scale by reacting carbon monoxide and sodium formate. It is an ionic compound that is formed when hydrogen atoms bond with carboxylic acid functional groups. Methanoic acid is a common chemical produced in industry. It is mainly made from carbon monoxide or methanol by heating it with sodium hydroxide and then reacting it with methyl formate. It is also manufactured from acetic acid. It is used as a reagent in the dyeing of natural and synthetic fibers, feed preservation, leather tanning, and commercial cleaning products. Formic Acid is also an ingredient in hydraulic fracturing, which uses a fluid to break up rock formations underground that hold oil and natural gas. This process increases the production of these resources. It is hazardous and skin irritant, so workers should be carefully monitored when in contact with it. Inhalation or skin/eye contact can cause blisters, burns, or a severe reaction. Exposure to high concentrations can lead to kidney damage. Methanoic acid is the simplest carboxylic acid and it can be found naturally in various sources like bees, ants stings and it is also a valuable synthetic organic reagent. It is commonly used in food preservatives, antimicrobial agents, and artificial flavorings. Ant stings produce methanoic acid through the distillation of their venom. Formic Acid can be made industrially from the base-catalyzed reaction of CO with methanol to produce methyl formate which is then hydrolyzed to form methanoic acid. Methanoic acid is a weak acid which dissociates partially in an aqueous solution. This property makes it dangerous for people to handle. It is also a corrosive chemical which can burn any part of the body if it comes in contact with the skin. Some of the symptoms of ingestion include pain, watering eyes, twitching eyelids, sensitivity to light, inflammation and burns. It can also cause respiratory problems. Hence, it is not recommended for anyone to handle it. Major applications for Naphthenic Acid include lubricants and greases, which are used extensively in the transportation and aviation industries. The molecular weight of a chemical compound is the amount of chemical substance present in a given quantity. It is a dimensionless number computed from the atomic weights of each element in the formula and then multiplied by the total weight of the chemical. It has a number of industrial applications, including dyeing natural and synthetic fibers, leather tanning, and the production of commercial cleaning products. It readily reacts with alcohols, forming esters. It is also a good reducing agent for sodium and potassium dichromate. Inhalation of fumes from diluted Formic Acid can cause irritation to the mucous membranes. Skin contact can result in blisters and burns. The Lewis structure of a molecule is a two-dimensional diagram that shows the bonding electron pairs between atoms. It also depicts lone pairs of electrons on an atom if they exist. Formic Acid has a total of 18 valence electrons. It has one valence electron on each of the hydrogen atoms, four on the carbon atom, and six on the oxygen atoms. It has four sigma covalent bonds. There are also eight nonbonding electrons. Each sigma covalent bond uses up eight of the 18 available electrons. This leaves 10 electrons that are not used for bonding.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
July 2023
Categories
All
|
 RSS Feed
RSS Feed
